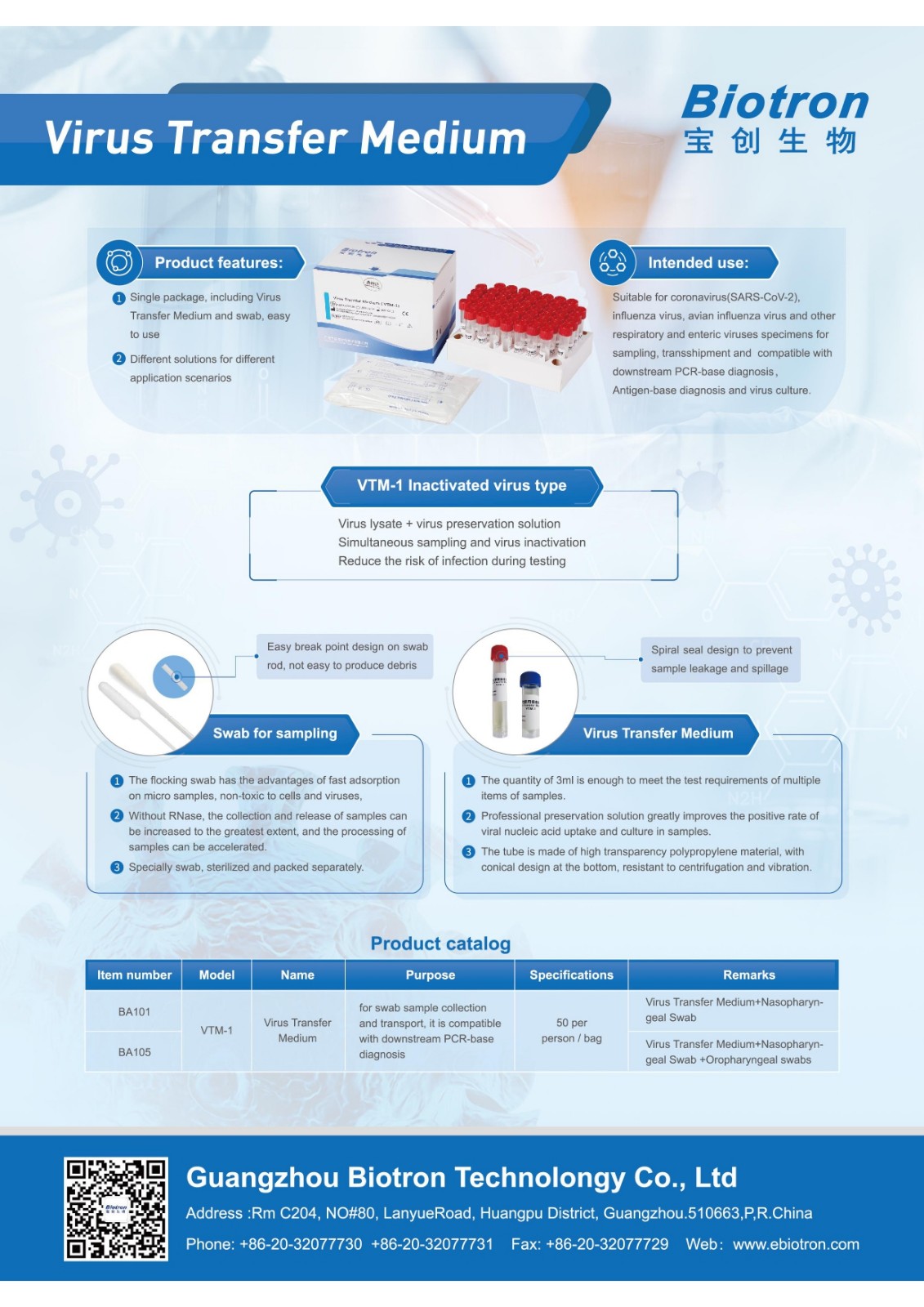
The new coronavirus is raging around the world. The nucleic acid detection of the new coronavirus is a key link in the prevention and control of the epidemic. How to efficiently and safely sample, transport, and detect nucleic acid has become a concern of frontline medical workers. Biotron has succeeded in developing antibodies and nucleic acids After the detection kit, it is committed to solving the difficulties in the initial stage of virus sampling, and launched a series of virus sampling/cultivation tubes, so as to realize the whole process and multi-faceted solution from sampling to detection.