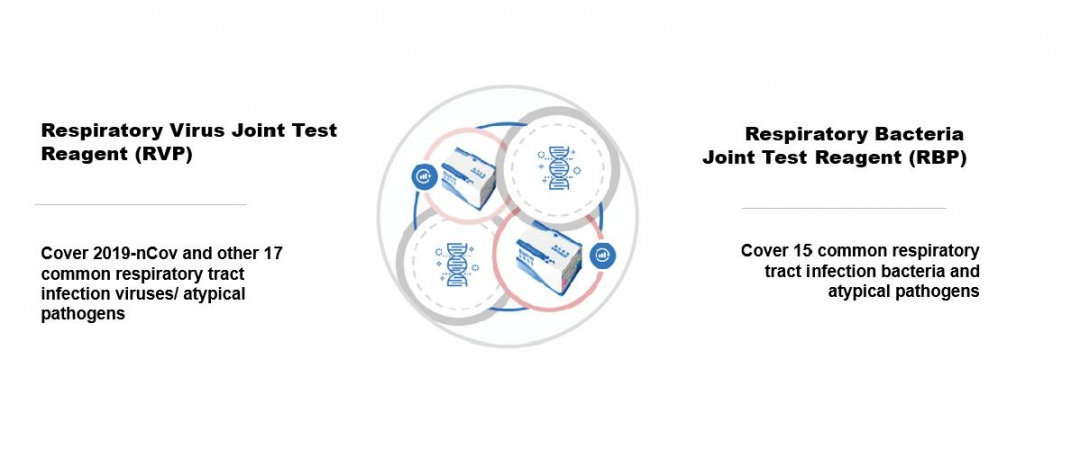
The prevention and control of the new coronavirus epidemic is at a critical juncture. The WHO 2019 New Coronavirus Guidelines and China's fifth edition of the "New Coronavirus Infection Pneumonia Diagnosis and Treatment Plan" both emphasize that the upper and lower respiratory tracts should be collected during laboratory examinations of suspected patients. , Blood, and fecal specimens are tested for nCoV through Real-time PCR. The results can be used for rapid identification, diagnosis, or judgment of cure.
Therefore, the development and application of high-quality, fast, accurate, low-cost, easy-to-operate, and versatile diagnostic reagents not only has important practical significance for the current 2019-nCoV epidemic control, but also for long-term monitoring and diversion of overall respiratory tract infections,diagnosis and treatment have long-term guiding significance.